Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
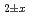
Watanabe, Masashi; Kato, Masato
Frontiers in Nuclear Engineering (Internet), 1, p.1082324_1 - 1082324_9, 2023/01
Since the oxygen potential and the oxygen coefficient of UO have a significant impact on fuel performance, many experimental data have been obtained. However, experimental data of the oxygen potential and the oxygen diffusion coefficient in the high temperature region above 1673 K are very limited. In the present study, we aimed to obtain these data and analyze them by defect chemistry. The oxygen potentials and the oxygen chemical diffusion coefficient of UO
have a significant impact on fuel performance, many experimental data have been obtained. However, experimental data of the oxygen potential and the oxygen diffusion coefficient in the high temperature region above 1673 K are very limited. In the present study, we aimed to obtain these data and analyze them by defect chemistry. The oxygen potentials and the oxygen chemical diffusion coefficient of UO were measured by the gas equilibrium method in the near stoichiometric region at temperatures ranging from 1673 to 1873 K. A data set of oxygen potentials was made together with literature data and analyzed by defect chemistry. The oxygen potential of UO
were measured by the gas equilibrium method in the near stoichiometric region at temperatures ranging from 1673 to 1873 K. A data set of oxygen potentials was made together with literature data and analyzed by defect chemistry. The oxygen potential of UO was determined as a function of O/U ratio and temperature, and an equation representing the relationship was derived. The oxygen chemical diffusion coefficient values obtained in this study were reasonably close to the literature values. The oxygen partial pressure dependence of the oxygen chemical diffusion coefficients was predicted from the evaluated results of the oxygen potential data, but no clear dependence was observed.
was determined as a function of O/U ratio and temperature, and an equation representing the relationship was derived. The oxygen chemical diffusion coefficient values obtained in this study were reasonably close to the literature values. The oxygen partial pressure dependence of the oxygen chemical diffusion coefficients was predicted from the evaluated results of the oxygen potential data, but no clear dependence was observed.
Sakai, Seiji; Naramoto, Hiroshi; Xu, Y.; Priyanto, T. H.; Lavrentiev, V.; Narumi, Kazumasa
Materials Research Society Symposium Proceedings, Vol.788, p.L11.49.1 - L11.49.6, 2004/00
Mixture films between cobalt and C (CoxC
(CoxC , x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C
, x = 0.5-700) were prepared by co-evaporation technique under UHV conditions. The composition-dependent features of dilatation and downshift of Raman peak suggest that the mixtures are composed of cobalt particles and a C -based phase in which certain number of cobalt atoms are coordinated with C
-based phase in which certain number of cobalt atoms are coordinated with C molecules. It is deduced that the equilibrated number of cobalt atoms in the C
molecules. It is deduced that the equilibrated number of cobalt atoms in the C -based phase is 4 atoms per C
-based phase is 4 atoms per C molecule and in which an electron transfer occurs from a cobalt atom to a C
molecule and in which an electron transfer occurs from a cobalt atom to a C molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x
molecule. The evaluation of their temperature dependences reveals that: (1) The mixtures with x 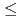 4 are thermally activated-type, and their conductive nature can be attributed to the C
4 are thermally activated-type, and their conductive nature can be attributed to the C -based phase. Further (2) the mixtures between 4
-based phase. Further (2) the mixtures between 4  x
x  60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x
60 are also thermally activated, however, the electron hopping process between the isolated cobalt particles is supposed to be operative (variable-range-hopping). And (3) in the mixtures with x  60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
60 which corresponds to a percolation threshold for cobalt particles, the system changes into metallic conduction.
Tamada, Masao; Asano, Masaharu; Yoshida, Masaru; Kumakura, Minoru
Radiation Physics and Chemistry, 36(6), p.866 - 867, 1990/00
no abstracts in English
; ; ;
Journal of Nuclear Materials, 151, p.90 - 94, 1987/00
Times Cited Count:3 Percentile:74.91(Materials Science, Multidisciplinary)no abstracts in English
; ; ; Katano, Y.; Katsuta, Hiroji
Journal of Nuclear Materials, 141-143, p.392 - 395, 1986/00
Times Cited Count:3 Percentile:40.87(Materials Science, Multidisciplinary)no abstracts in English
; Furukawa, Kazuo
First Inter.Symp.on Molten Salt Chem.Technol.,J-315, p.449 - 452, 1983/00
no abstracts in English
 -methylstyrene
-methylstyrene; *; *
Polym.J., 3(6), p.762 - 763, 1972/06
Times Cited Count:6no abstracts in English
 C; Effect on electrical resistivity and thermal conductivity
C; Effect on electrical resistivity and thermal conductivity;
Journal of Nuclear Materials, 45(1), p.79 - 81, 1972/01
Times Cited Count:2no abstracts in English
; *; *; *
Radiat.Eff., 1, p.109 - 116, 1969/00
no abstracts in English
; *; *; *
Radiation Effects, 1, p.109 - 116, 1969/00
no abstracts in English
; ; *; *; *
Bulletin of the Chemical Society of Japan, 41(5), p.1261 - 1263, 1968/00
Times Cited Count:7no abstracts in English